Un supercapacitor este cunoscut si sub denumirea de ultracapacitor sau condensator electrolitic cu doua straturi. Acesta este un tip de condensator care poate stoca o cantitate mare de energie, de obicei de 10 pana la 100 de ori mai multa energie comparativ cu condensatoarele electrolitice.
Dezasamblarea unui supercondensator

Cum functioneaza un supercapacitor?
Supercapacitorii utilizeaza doua mecanisme diferite pentru a stoca energia electrica, capacitatea electrostatica bazata pe stratul dublu electric si pseudocapacitatea electrochimica. Acest lucru inseamna ca supercapacitorii combina proprietatile condensatoarelor si bateriilor electrice intr-un singur dispozitiv.
Supercapacitorii sunt formati din doi electrozi, un electrolit, membrana separatoare, iar performantele lor difera in functie de caracteristicile materialelor folosite. Acestia nu functioneaza pe baza unor reactii chimice, precum in cazul bateriilor conventionale, ci pe baza generarii unor campuri electrostatice. Acesti condensatori sunt polarizati, deci trebuie conectati in mod corect, la fel ca si condensatoarele electrolitice.
Dezavantajele utilizarii supercapacitorilor
Principalele impedimente in calea utilizarii supercapacitorilor sunt costul ridicat si densitatea scazuta de energie ce caracterizeaza aceste dispozitive. Comparativ cu bateriile Li-ion obisnuite, supercapacitorii actuali nu pot stoca o cantitate mare de energie electrica si nu o pot mentine prea mult timp.
Din acest motiv supercapacitorii, in forma lor actuala, nu pot asigura o incarcare cu energie electrica de lunga durata a autovehiculelor electrice.
Unde sunt utilizati supercondensatorii?
Supercondensatoarele sunt utilizate in aplicatii care necesita multe cicluri rapide de incarcare / descarcare, mai degraba decat stocarea compacta pe termen lung a energiei – in automobile, autobuze, trenuri, macarale si lifturi, unde sunt utilizate pentru franare regenerativa, stocare de energie pe termen scurt sau livrare de energie in modul rafala. Unitatile mai mici sunt utilizate ca rezerva de energie pentru memorie statica cu acces aleatoriu (SRAM).
Structura interna a supercapacitorului
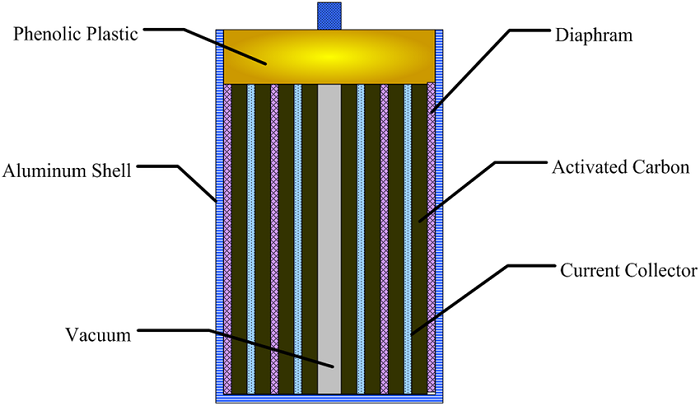
Electrolitul formeaza o legatura ionica conductiva intre cei doi electrozi, care ii deosebeste de condensatorii electrolitici conventionali in care exista intotdeauna un strat dielectric si asa-numitul electrolit, de exemplu, MnO2 sau polimer conducator, este de fapt parte a celui de-al doilea electrod (catodul, sau mai corect electrodul pozitiv). Supercondensatorii sunt polarizati prin proiectare cu electrozi asimetrici sau, pentru electrozi simetrici, de un potential aplicat in timpul fabricatiei.
Top 12 producatori de supercapacitori
- Cellergy (USA)
- Ioxus (USA)
- Maxwell Technologies (USA)
- Murata Manufacturing (Japan)
- Nanoramic Laboratories (USA)
- Nec Tokin (Japan)
- Nippon Chemi-Con (Japan)
- Panasonic (Japan)
- Paper Battery Company (USA)
- Skeleton Technologies (Estonia)
- Yunasko (UK)
- ZapGo (UK)
Bibliografie:
https://www.stiintaonline.ro/
https://wikicro.icu/
https://www.researchgate.net/
https://www.exponent.com/
https://www.thomasnet.com/
https://www.techspot.com/


The name supercaps is also used for quite small capacitors (but, with a relatively high capacitance for their size) which are used to provide back up power to memory, real time clocks etc. when the main power is switched off. They are also referred to as EDL capacitors, Electric Double Layer capacitors. Charging-discharging occurs in the ion absorption layer which is formed on the surfaces of the positive and negative electrodes. Manufacturers of this type of capacitor use two types of electrolyte. One is water-soluble and the other is non-watersoluble.
The latter can withstand higher voltage per cell. This type of capacitor thus uses
special techniques in order to achieve such high capacitance values in a compact
package. Just like an electrolytic capacitor it is polarized.
Have they started replacing car batteries yet? That would be grand!
This idea is still on hold, probably due to the cost.
We need to harness the energy of nature.. just imagine if we could use it’s full potential energy today? We can cure our problems if we move the tree of greed aside seeing the vastness of electricity in it’s raw form as in the case of lighting. Earth’s natural production of electricity.
Yes, car manufacturers would then produce cars that could accelerate to 400km/h in 2 seconds and speed limits being reduced! Funny old world, huh!
When Ford produced his first vehicle, I recall reading somewhere it was battery powered? It didn’t have a catalytic converter had a fuel economy of nearly a 100 miles per gallon. That was before Paul Getty got involved with his monopoly of crude oil?
Ford’s first car battery powered, had an economy of 100mpg? Sorry I don’t understand! Part of its economy was due to the fact it didn’t weight 2 tons and travel at 100mph, if we drove practical and sensible vehicles their carbon footprint and energy consumption would be much reduced, be it gas, petrol, diesel, electric .
Multumesc pentru acest articol informativ despre supercapacitoare! Am aflat multe lucruri interesante despre acest dispozitiv si cum functioneaza el pentru a stoca energia electrica. Cu siguranta va fi o solutie excelenta pentru aplicatiile care necesita cicluri rapide de incarcare si descarcare. Totusi, din pacate din cauza costului ridicat si a densitatii scazute de energie, nu poate inlocui complet bateriile conventionale in cazul autovehiculelor electrice. Dar sunt convinsa ca in viitor, odata cu progresul tehnologic, supercapacitoarele vor deveni o solutie eficienta si accesibila pentru stocarea energiei. Felicitari pentru munca depusa in realizarea acestui material de calitate!
Supercondensatoarele sau supercapacitorii reprezinta viitorul in domeniul stocarii energiei electrice. Nu doar ca pot stoca cantitati mari de energie, insa sunt si mai eficienti in termeni de viteza de incarcare si descarcare. De asemenea, sunt utilizati in mai multe domenii, inclusiv in industria auto. Chiar daca mai exista dezavantaje in ceea ce priveste densitatea de energie, se fac constant cercetari pentru a imbunatati acest aspect. Cu siguranta, supercapacitorii vor revolutiona lumea tehnologiei energetice!